

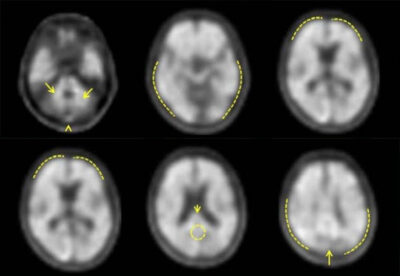
An Amyloid PET scan is a key step on the pathway to treatment for
When memory loss begins to affect daily life, knowing the cause is critical. A brain scan, also known as a PET Scan, with Neuraceq® gives your doctor information that can help assess your condition, assisting in the diagnosis of Alzheimer's Disease or ruling it out.




Neuraceq® - Indications, Use and Important Safety Information
PRODUCT INDICATIONS AND USE: NEURACEQ is a radioactive diagnostic drug indicated for positron emission tomography (PET) of the brain to estimate amyloid beta neuritic plaque density in adults with cognitive impairment for: Evaluation of Alzheimer’s disease (AD) and other causes of cognitive decline, and selection of patients who are indicated for amyloid beta-directed therapy as described in the prescribing information of the therapeutic products.
NEURACEQ IMPORTANT SAFETY INFORMATION:
CONTRAINDICATIONS: None
WARNINGS AND PRECAUTIONS:
ADVERSE REACTIONS:
USE IN SPECIFIC POPULATIONS:
OVERDOSAGE:
In the event of administration of a radiation overdose with NEURACEQ, the absorbed organ dose to the patient should be reduced by increasing elimination of the radionuclide from the body by inducing frequent micturition. Prior to NEURACEQ administration, please read the full Prescribing Information for additional Important Safety Information.
SUSPECTED ADVERSE REACTIONS: please report to: https://www.fda.gov/safety/medwatch-fda-safety-information-and-adverse-event-reporting-program
NEURACEQ® (florbetaben F18 Injection) is a FDA approved imaging medicine used during a brain scan called positron emission tomography (PET). It helps doctors to see whether certain protein deposits, called amyloid plaques, are present in the brain.
This scan is used in adults who are experiencing memory or cognition problems to help:
Image interpretation errors have been observed. There may be differences or errors when evaluating the presence of amyloid plaque in the brain using NEURACEQ®. Certain factors—such as significant brain shrinkage or movement during the scan—can make the images harder to read and may affect how clearly different parts of the brain can be seen.
Your specialist reads the NEURACEQ® image without taking into account your symptoms or medical history to avoid presumptions. In cases where it is difficult to tell exactly where a signal comes from in the brain, your healthcare providers may compare the NEURACEQ® scan with another type of brain image (magnetic resonance imaging or computer tomography) to better understand the findings.
NEURACEQ® uses a small amount of radiation, which adds to a person’s total lifetime exposure to radiation. Over time, higher amounts of radiation exposure may increase the risk of cancer. Health care professionals take special precautions to handle this medication safely and reduce unnecessary radiation exposure for both patients and staff. You are encouraged to drink plenty of fluids before and after the scan and to urinate often afterward.
There is limited information about the use of NEURACEQ® during pregnancy. It is not known whether this scan could affect an unborn baby because studies in pregnant women and animals in reproduction have not been done. NEURACEQ® contains a small amount of radiation. All imaging medicines that use radiation may pose some risk to an unborn baby, depending on how far along the pregnancy is and how much radiation is involved.
If you are pregnant or think you might be pregnant, talk with your health care provider before having this scan. They will discuss the potential risks and benefits with you. It is important to know that all pregnancies have some natural risk. In the general population, about 2–4% of babies are born with a major birth defect, and about 15–20% of pregnancies end in miscarriage.
It is not known whether NEURACEQ® passes into breast milk, how it might affect a breastfed baby or whether it could impact milk production. To reduce any possible radiation exposure to a nursing infant, mothers are advised to pump and discard breast milk for 24 hours after receiving NEURACEQ® to minimize radiation exposure. Breastfeeding can be resumed after that time. When making decisions about breastfeeding, both the benefits of breastfeeding and the mother’s medical need for the scan should be considered.
NEURACEQ® has not been studied in children. Its safety and effectiveness are not known for pediatric patients.
NEURACEQ® has been studied in adults aged 65 and older, including those over age 75. Studies did not show differences in safety or effectiveness between older adults and younger adults.
If too much NEURACEQ® is given by mistake, your body may clear it more quickly by drinking fluids and urinating often. This helps reduce radiation exposure.
There are strict laws on the use, handling and disposal of radiopharmaceutical products. NEURACEQ® is given only by trained and qualified health care providers who have specific experience working with imaging medicines that contain small amounts of radiation. These providers are licensed and approved by appropriate regulatory authorities to use these products safely.
The most common side effects (incidence ≥ 1%) were injection site pain, injection site redness, or irritation at the injection site.
This is not a complete report of risk information for NEURACEQ®. Talk to your healthcare provider about any side effects that concern you. You are encouraged to report negative side effects of prescription drugs to FDA: visit www.fda.gov/medwatch or call 1-800-FDA-1088 OR Lantheus Biosciences at 1-833-491-2524 OR PHV@lantheus.com.
Please see full Prescribing Information, including Important Facts for NEURACEQ®.
© 2026 Lantheus. All rights reserved.
NEURACEQ® is a radioactive diagnostic drug indicated for positron emission tomography (PET) of the brain to estimate amyloid beta neuritic plaque density in adults with cognitive impairment for:
Please see full Prescribing Information for NEURACEQ®
Image interpretation errors have been observed. Errors may occur in the estimation of brain amyloid beta neuritic plaque density during NEURACEQ® image interpretation. The use of clinical information in the interpretation of NEURACEQ® images has not been evaluated and may lead to an inaccurate assessment. Severe brain atrophy as well as motion artifacts that result in image distortion may limit the ability to distinguish gray and white matter on a NEURACEQ® scan. Perform image interpretation independently of the patient’s clinical information. For cases where there is uncertainty as to the location of cortical signal, use co-registered anatomical imaging to improve localization of signal.
NEURACEQ® contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk of cancer. Ensure safe drug handling to protect patients and health care providers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
Most common adverse reactions (incidence ≥1%) were injection site pain, injection site erythema, and injection site irritation.
Lactation: Temporarily discontinue breastfeeding. A lactating woman should pump and discard breast milk for 24 hours after NEURACEQ® administration.
To report suspected adverse reactions of NEURACEQ®, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. or Lantheus Biosciences Ltd. at 1-833-491-2524 or phv@lantheus.com.
© 2026 Lantheus. All rights reserved.
The information contained in Neuraceq.com/hcp is intended for healthcare professionals in the United States only. If you are a US healthcare professional, click the "Continue" button below.
Yes, I am a US healthcare professional and would like to continue.
We use cookies to help you navigate efficiently and perform certain functions. You will find detailed information about all cookies under each consent category below.
The cookies that are categorized as "Necessary" are stored on your browser as they are essential for enabling the basic functionalities of the site.... Show more
We also use third-party cookies that help us analyze how you use this website, store your preferences, and provide the content and advertisements that are relevant to you. These cookies will only be stored in your browser with your prior consent.
You can choose to enable or disable some or all of these cookies but disabling some of them may affect your browsing experience.
Always Active
Necessary cookies are required to enable the basic features of this site, such as providing secure log-in or adjusting your consent preferences. These cookies do not store any personally identifiable data.
Functional cookies help perform certain functionalities like sharing the content of the website on social media platforms, collecting feedback, and other third-party features.
Analytical cookies are used to understand how visitors interact with the website. These cookies help provide information on metrics such as the number of visitors, bounce rate, traffic source, etc.
Performance cookies are used to understand and analyze the key performance indexes of the website which helps in delivering a better user experience for the visitors.
Advertisement cookies are used to provide visitors with customized advertisements based on the pages you visited previously and to analyze the effectiveness of the ad campaigns.